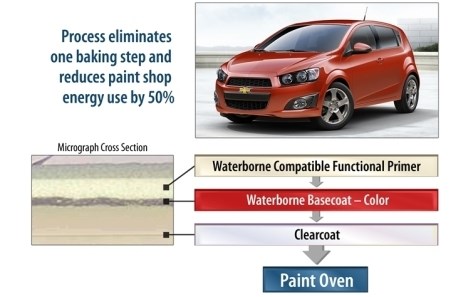
Chevy Sonic Gets Eco-Friendly Paint Job
Thanks to this system, as well as other recent shop enhancements designed to minimize energy use and paint-solvent emissions, Orion Assembly’s paint facility will reduce greenhouse gas emissions by approximately 80,000 metric tons each year at full production, cut solvent emissions by about 108,000 lbs. annually, slice $40 of manufacturing costs from each vehicle and use 50 percent less process energy per vehicle—all while being heated by natural and landfill gas instead of more emissions-intensive coal-fired boilers.
Share
Related Content
-
Spatial Coating Thickness Measurement Solution for Prismatic Battery Cells
Coatmaster AG’s 3D noncontact coating thickness measurement system employs ATO technology to provide spatial measurement of coating thickness.
-
Solar-Powered Photonic Cooling Enables Energy-Saving Coating
Passive cooling technology can reduce interior temperatures 5-13°C, offering weight, cost and CO2 benefits for cars, construction, aircraft and more.
-
Online Energy Savings Calculator Promotes Energy Efficiency
An online energy savings calculator from AkzoNobel aims to help customers determine how to reduce energy usage.












